Introduction
Crop diseases constitute a major threat in agricultural production systems that deteriorate yield quality and quantity at production, storage, and transportation level. At farm level, reports on yield losses, due to crop diseases, are very common. Furthermore, crop diseases pose significant risks to food security at a global scale. Timely identification of crop diseases is a key aspect for efficient management. Crop diseases may be provoked by various kinds of bacteria, fungi, pests, viruses, and other agents. Disease symptoms, namely the physical evidence of the presence of pathogens and the changes in the crops’ phenotype, may consist of leaf and fruit spots, wilting and color change, curling of leaves, etc. Historically, disease detection was conducted by expert agronomists, by performing field scouting. However, this process is time-consuming and solely based on visual inspection. Recent technological advances have made commercially available sensing systems able to identify diseased crops before the symptoms become visible.
Reliable diagnosis of diseases and pests in the early stages of crop production is highly desirable to reduce significant production and economic losses. The main purpose of crop pest diagnosis is to assess crop health and determine the cause of the disease. However, one of the major challenges is the difficulty in determining the physical, chemical, and biological changes in crops during the asymptomatic stage of infection. Another challenge is that it is difficult to work in a timely and economical manner. Crop diseases result in significant production and economic losses to agriculture around the world. Monitoring the health of crops and trees and detecting diseases is important for sustainable agriculture. As far as we know, there are no commercially available sensors that evaluate the condition of trees in real time. Scouting is the most widely used mechanism for monitoring tree stress today. This is a costly, labor-intensive and time-consuming process. Molecular techniques such as the polymerase chain reaction are used to identify crop diseases that require detailed sampling and processing procedures. Initial information for crop health and disease detection should facilitate disease management using appropriate management strategies such as pesticide application, fungicide application, and vector control through disease-specific chemical application.
There is a need for fast and reliable diagnostic methods that can be used in the field to detect crop diseases at the asymptomatic stage. Indirect methods that rely on imaging techniques and the profile of VOCs emitted from abundant crops can meet these needs. For example, for early detection of stress, spectroscopic imaging techniques used both in the field and in cultivated greenhouse crops show satisfactory classification accuracy. However, some changes and improvements are still needed, including temporary consequences. Biosensors that use phage display and biophotonics have been reported to detect infections immediately, but must be corrected, improved, and properly validated before being used in the field. Therefore, the purpose of this study is to organize, examine and compare the characteristics of the three methods for detecting crop diseases.
Crop Disease Technologies
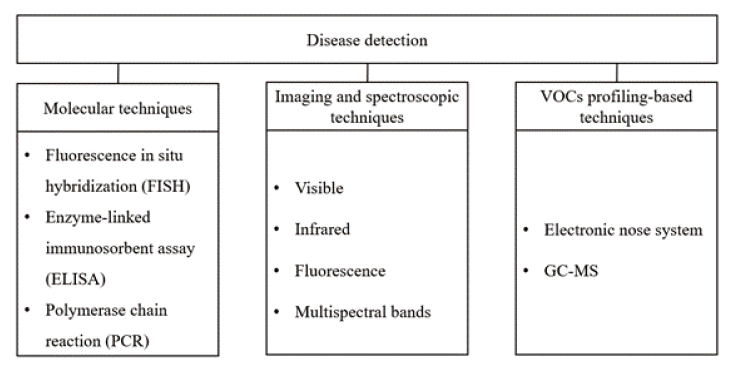
In order to solve the problems of these crop diseases, various detection techniques have been developed. Figure 1 shows a representative crop disease detection technique. Disease detection techniques are largely divided into three types: Molecular techniques, Imaging and spectroscopic techniques, and VOCs profiling-based techniques. Molecular techniques are direct detection methods, including polymerase chain reaction (PCR), fluorescence in-situ hybridization (FISH) and serological technologies such as enzyme-linked immunosorbent assay (ELISA). Spectroscopic and imaging techniques are unique disease monitoring methods used to detect disease and stress caused by a variety of crop and tree factors. Current research activities are directed towards the development of such techniques for creating practical tools for large-scale real-time disease monitoring under field conditions. Various spectroscopic and imaging techniques have been studied to detect symptomatic and asymptomatic crop diseases. Several methods include fluorescence imaging (Bravo et al., 2004; Chaerle et al., 2007), multispectral imaging (Qin et al., 2009), Infrared spectroscopy (Spinelli et al., 2006; Purcell et al., 2009) Visible and Multiband Spectroscopy (Yang et al., 2007; Delalieux et al., 2007; Chen et al., 2008). VOCs profiling-based techniques are largely divided into electronic nose system and GC-MS. Typical indirect methods detect morphological changes, transpiration rate changes, and volatile organic compound (VOC) profiles. These correspond to fluorescence imaging, hyperspectral technology, and gas chromatography-mass spectrometry (GC-MS) technology.
Molecular techniques of crop disease detection
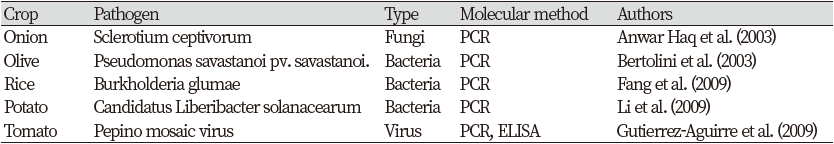
In recent years, molecular technology for detecting crop diseases has been well established. Using molecular methods to diagnose crop diseases offers many advantages to the diagnostician over traditional methods. For example, they can enable the identification of morphologically similar species. Molecular tools not only improve the effectiveness, accuracy and speed of diagnosis, but their common technical foundation is especially when resources are limited and traditional skills are difficult to maintain. It offers additional benefits. The sensitivity of molecular technology is the minimum amount of microorganisms that can be detected in a sample.
Lopez et al. (2003) reported that the sensitivity of molecular techniques for detecting bacteria ranges from 10 to 106 colony forming units /mL. Molecular techniques commonly used to detect disease are ELISA and PCR (PCR and real-time PCR). Other molecular technologies include immunofluorescence (IF), flow cytometry, fluorescence in situ hybridization (FISH), and DNA microarrays. Disease detection by ELISA involves injecting a protein (antigen) from a microorganism associated with a crop disease into an animal that produces antibodies to that antigen. These antibodies are extracted from the animal's body and used for antigen detection with fluorescent dyes and enzymes. In the presence of disease-causing microorganisms (antigens), the sample fluoresces, confirming the presence of disease in a particular crop. The presence of specific bands in gel electrophoresis confirms the presence of organisms responsible for crop diseases. There are many studies on disease detection using molecular technology. Efforts are underway to improve the efficiency of these technologies.
Spectroscopic and imaging techniques for disease detection
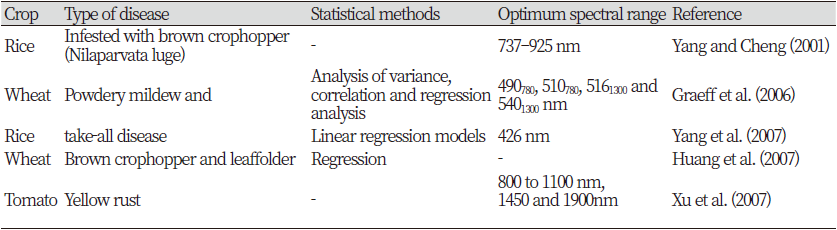
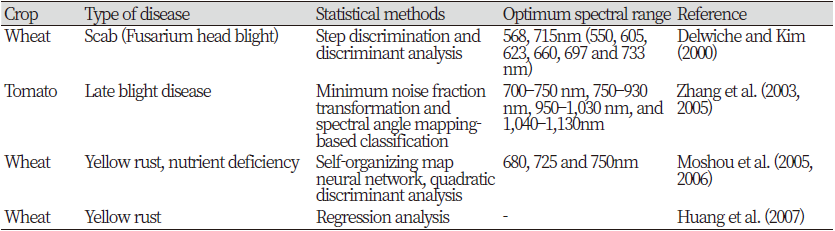
Recent developments in agricultural technology have lead to a demand for a new era of automated non-destructive methods of crop disease detection. It is desirable that the crop disease detection tool should be rapid, specific to a particular disease, and sensitive for detection at the early onset of the symptoms (Lopez et al., 2003). The spectroscopic and imaging techniques are unique disease monitoring methods that have been used to detect diseases and stress due to various factors, in crops and trees. Current research activities are towards the development of such technologies to create a practical tool for a large-scale real-time disease monitoring under field conditions. Various spectroscopic and imaging techniques have been studied for the detection of symptomatic and asymptomatic crop diseases. Some the methods are: fluorescence imaging (Bravo et al., 2004; Moshou et al., 2005; Chaerle et al., 2007), multispectral or hyperspectral imaging (Moshou et al., 2004; Shafri and Hamdan, 2009; Qin et al., 2009), infrared spectroscopy (Spinelli et al., 2006; Purcell et al., 2009), fluorescence spectroscopy (Marcassa et al., 2006; Belasque et al., 2008; Lins et al., 2009), visible/multiband spectroscopy (Yang et al., 2007; Delalieux et al., 2007; Chen et al., 2008), and nuclear magnetic resonance (NMR) spectroscopy (Choi et al., 2004). Hahn (2009) reviewed multiple methods (sensors and algorithms) for pathogen detection, with special emphasis on postharvest diseases.
As fluorescence spectroscopy, visible and infrared spectroscopy are used as a fast, non-destructive, and cost-effective method for detecting crop diseases. This is a rapidly evolving technology used in a variety of applications (Ramon et al., 2002; Delwiche and Graybosch, 2002). Studies have also been conducted on the detection of stress, damage and disease in crops using this technique (Spinelli et al., 2006; Naidu et al., 2009). The visible and infrared regions of the electromagnetic spectrum are known to provide the greatest information on the physiological stress levels of crops (Muhammed, 2005; Xu et al., 2007) and these wavelengths specific to disease. You can use some of the bands. To discover crop diseases (West et al., 2003), before symptoms are visible. Visible spectroscopy is commonly used in combination with infrared spectroscopy to detect crop diseases (Bravo et al., 2003; Huang et al., 2004; Larsolle and Muhammed, 2007).
Profiling of crop volatile organic compounds for disease detection
The volatile organic compounds (VOCs) released from crops and trees account for about two-thirds of the total VOC emissions present in the atmosphere (Guenther, 1997). There are many factors that affect the volatile metabolic profile of a crop or tree. VOCs emitted by crops depend on a variety of physicochemical factors such as humidity, temperature, light, soil conditions, fertilization, and biological factors such as crop growth and development stages, the presence of insects and other herbs. (Vallat et al., 2005; Vuorinen et al., 2007). Physicochemical factors directly or indirectly affect the physiological state of the crop, thereby affecting the VOC profile of the crop. Volatile substances in these crops affect the relationship between crops and other organisms, including pathogens (Vuorinen et al., 2007). For example, acetaldehyde released from the leaves of young poplar trees is controlled by the transfer of ethanol to the leaves by transpiration (Kreuzwieser et et. Et al. 2001).
The electronic nose system consists of a series of gas sensors that react with a variety of organic compounds. Because each sensor has a specific sensitivity, the sensitivity of a set of sensors can be used to identify different compounds present in the atmosphere. Electronic nose systems are used in a variety of applications. They have been used to determine food quality (Evans et al., 2000; Di Natale et al., 2001; Zhang et al., 2008a, b)., 2001; Dragonieri et al., 2007), Detection of microorganisms in food (Falasconi et al., 2005; Rajamäki et al., 2006; Balasubramanian et al., 2008; Concina et al., 2009). Applications of electronic nasal systems for identifying crop diseases are a relatively new area of application.
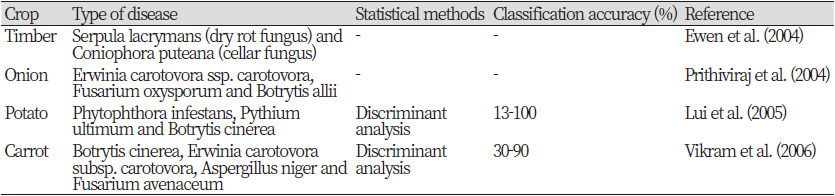
GC-MS is a commonly used technique for qualitative and quantitative analysis of volatile metabolites released from crops / trees under a variety of environmental and physiological conditions. GC-MS studies were performed to assess changes in volatile substances caused by bacterial or fungal infections in various foods (Table 5). Prithiviraj etc. (2004) Bacterial species (Erwinia carotovora causes Erwinia carotovora) and fungal species (Fusarium oxysporum and roots) using HAPSITE, a commercially available portable GC-MS device. We evaluated the variability of volatiles released from the bulbs of onions infected with Pectobacterium carotoides (Botrytis allii). This study showed that 25 volatile compounds released from onions (out of 59 consistently detected compounds) could be used to identify the disease based on VOC profiling. No statistical analysis was performed to determine the discriminating ability of the algorithm in classifying VOC profiles for disease detection, but model development and software development were recommended for this purpose.
A similar study of potato tubers inoculated with Erwinia carotovora subsp. carotovora, E. carotovora subsp. Atroseptica, Pythium ultimum, Phytophthora infestans, or Fusarium sambucinum using solid-phase microextraction (SPME) fibers and GC flame ionization detectors (FIDs) have shown potential for VOC profiling for disease detection (Kushalappa). et al., 2002). The amount of volatiles increased with increasing severity of the disease. The BPNN model was applied to classify volatile metabolite profiles for disease. The gas retention time (GC function) of the volatile compounds was used as input data and the two hidden layers were used for mutual validation. Mutual validation probabilities (using BPNN) were> 67% (67-75%) in all groups except potato tubers infected with Phytophthora. Unlike other studies, this study did not identify the specific compound that caused the VOC peak in the FID.
Conclusion
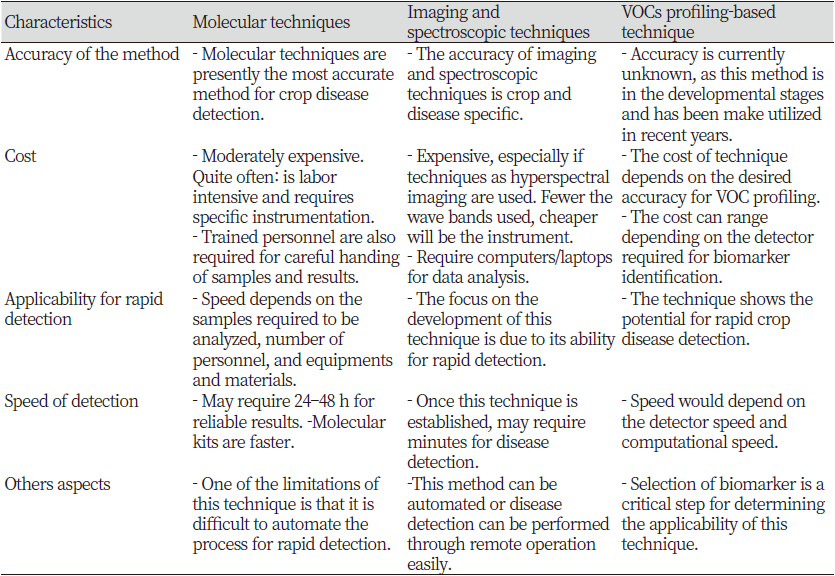
An overall comparison of the three key technologies is summarized in Table 6. Recent literature reports support the idea that volatile profiling and changes in spectral reflectance can be used for non-invasive field monitoring of crop diseases. Crops and trees release volatile organic compounds (VOCs) as a result of metabolic activity that occurs in buds, leaves, flowers and fruits. The VOC profile of each crop varies greatly depending on physiological conditions and species. Various factors affect the VOC profile of a particular crop or tree. This includes changes in the metabolism of crops that are affected by changes in the environment, the age of the crop, the stage of crop development, the stress of the crop, and the presence of the disease. One of the biggest problems when using crop airframe metabolites as an indicator of the presence of crop diseases is the natural change in VOC profile among crop species. Fluctuations in VOCs released by crops can hide changes due to stress and illness. Therefore, unlike VOCs produced by environmental and nutritional stress, it is necessary to identify various volatile biomarkers that are specific to a particular crop or disease. Real-world applications require the development of robust and reliable systems for real-time monitoring of crop diseases. Similar to the VOC profile of crops, environmental conditions affect the spectral reflectance of an object (Griffin and Burke, 2003). Therefore, you need to identify the appropriate approach to solving this problem. A possible way to overcome this problem is to identify the wavelength range or exponent that is not only sensitive to the disease of a particular crop, but is least affected by changes in environmental conditions. Autonomous robots can integrate imaging and VOC profiling techniques. This is because these technologies are well established for other industrial application areas. Once well established for specific disease detection applications, such methods can be integrated with autonomous agricultural vehicles to monitor crop diseases in real time.
This paper reviews and summarizes some of the non-invasive techniques used to detect crop diseases. The two main categories of non-invasive monitoring of crop diseases are (1) spectroscopy and imaging techniques and (2) basic volatile organic compound profiling techniques for recognizing crop diseases. Spectroscopy and imaging techniques include fluorescence spectroscopy, visible infrared spectroscopy, fluorescence imaging and hyperspectral imaging. Disease detection based on VOC profiles involves using analysis of electronic nasal or GC-MS-based volatile metabolites released from healthy and diseased crops as a tool for identifying diseases. It will be. I would. Some of the challenges of these technologies are influenced by (1) the background data of the resulting profile data, (2) optimization of the technology for specific crop diseases and (3) continuous automation. Monitor Technology automation. Crop diseases under actual field conditions. This study shows that methods for detecting these diseases have excellent potential for the ability to accurately detect crop diseases. Spectroscopic and imaging technologies can be integrated with autonomous agricultural vehicles to achieve superior crop disease control and control through reliable real-time crop disease detection.